Battery power is becoming a standard part of everyday life, whether for your child’s toy, cordless power tool, or electric vehicle. One of the most critical components of a battery is the internal electrolyte.
Today, we’re exploring battery electrolytes and how they work to power your electronics. Let’s dive in!

What Is the Battery Electrolyte?
The battery electrolyte is a liquid or paste-like substance, depending on the battery type. However, regardless of the type of battery, the electrolyte serves the same purpose: it transports positively charged ions between the cathode and anode terminals.
How Does a Battery Work?
A battery has three major components—the positive terminal (cathode), the negative terminal (and)e, and an electrolyte that separates the two. The electrolyte is a solution that allows electrically charged particles (ions) to pass between the two terminals (electrodes). By releasing the chemicals required for the reaction, the electrolyte comes in contact with the anode and cathode, converting stored energy into usable electrical energy. This reaction provides power to the connected device, whether a light, a vacuum, or an electric vehicle.
What Is the Battery Electrolyte Made Of?
Different types of batteries rely on various chemical reactions and electrolytes. For example, a lead-acid battery usually uses sulfuric acid to create the intended reaction. Zinc-air batteries rely on oxidizing zinc with oxygen for the reaction. Potassium hydroxide is the electrolyte in standard household alkaline batteries. The most common electrolyte in lithium batteries is a lithium salt solution such as lithium hexafluorophosphate (LiPF6).

If you remember your high school chemistry class, you’ll likely remember wearing safety goggles and other protective gear when handling chemicals. Chemicals you’d use to create chemical reactions in batteries are often hazardous, so take the proper precautions when working with batteries and their electrolytes.
Can You Add an Electrolyte To A Battery?
Yes, you can add electrolytes to a battery, but ONLY if it’s a non-sealed wet cell battery. Checking the levels in a wet cell battery is standard maintenance that should be done regularly.

The electrolyte in these batteries contains water and sulfuric acid. When properly functioning, a wet cell battery will only consume water. So, in this case, simply adding distilled water will help maintain the proper electrolyte levels.
If your battery is sealed or doesn’t consume the electrolyte while off-gassing, nothing needs to be added to it. The lack of off-gassing is one advantage of AGM or lithium-ion batteries, as they require very little maintenance once installed.
What Are the Chemicals in Lithium Batteries?
The composition of a lithium battery depends on the chemistry that creates the reaction and the type of lithium battery. Most lithium batteries use a liquid electrolyte, such as LiPF6, LiBF4, or LiClO4, in an organic solvent.

However, recent advances have enabled the creation of solid-state batteries using solid ceramic electrolytes, such as lithium metal oxides. Solid-state technology’s main advantage is that it eliminates the risk of leaking and flammability, a safety risk with liquid electrolytes.
Lithium hexafluorophosphate (LiPF6) is the most common lithium salt in lithium-ion batteries. This solution creates an incredibly stable environment for the lithium ions during charging and discharging.
How Lithium Batteries Work
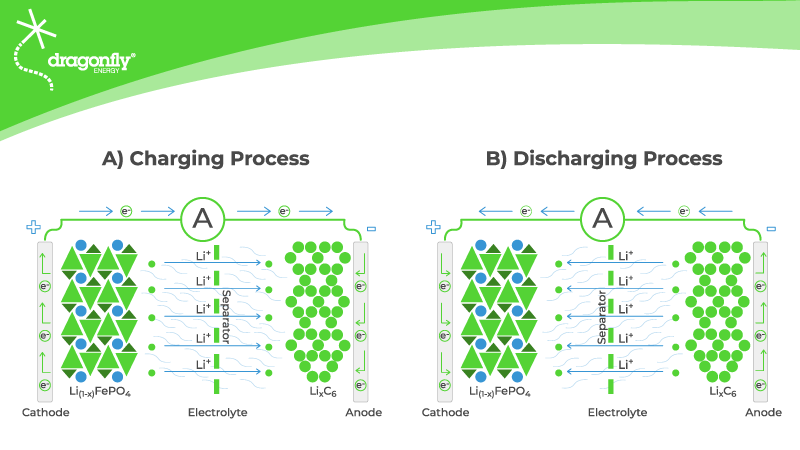
Lithium-ion batteries use charged lithium ions to create an electrical potential between the anode and cathode terminals. A thin layer of insulating material called a “separator” sits in the electrolyte solution between the two terminals. The separator allows the lithium ions to pass through while blocking the electrons and keeping the two electrodes apart. Lithium ions move through the separator from the positive side to the negative during charging. While discharging, the ions move in the opposite direction.
The movement of the lithium ions creates an electrical potential difference called “voltage.” When you connect your electronic devices to the battery, electrons (not lithium ions) flow through your device and power it.
Is Lithium Battery Electrolyte Safe?
The electrolytes in lithium batteries are safe. However, in the early days of lithium batteries, thermal runaway was a more prevalent issue when the batteries caught fire. The fires were mainly due to solvents in the lithium cells overheating, getting punctured, or overcharging.
As technology advances, new options are becoming available to improve the safety of lithium batteries. For example, Battle Born’s proprietary battery management system (BMS) will shut down the battery cells if it detects unsafe conditions, resulting in one of the safest batteries on the market.

A Critical Component of Your Battery
Battery electrolytes are critical components in all types of batteries. In most cases, you’ll probably never even think about them. However, understanding how they work can help extend the life of your battery.